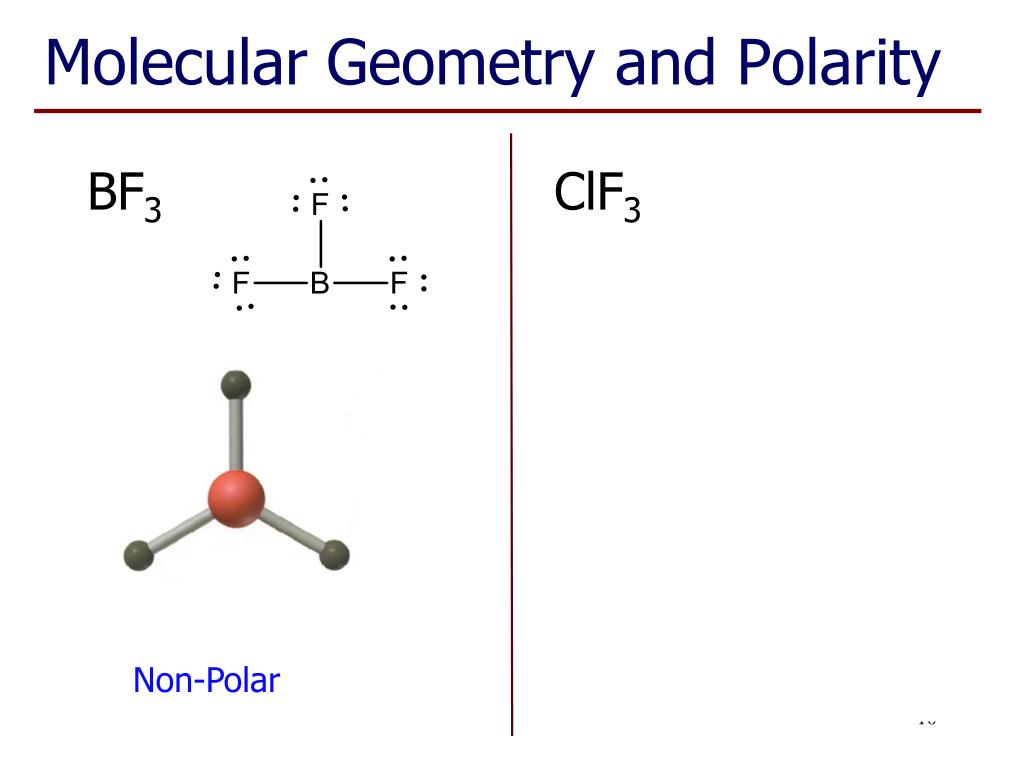
That leaves one equatorial, and 2 axial, sites where the Cl-F bonds can go, which leaves you with a T-shaped geometry, rather than a trigonal planar structure. This molecule has two lone pairs and three bound pairs, according to the ClF3 Lewis structure. Therefore, the interactions are lowest in the equatorial environment, so this is where the lone pairs go. ClF3 has a T-shaped molecular geometry and trigonal bipyramidal electron geometry. In an equatorial environment however, only 2 bonds/lp's are at a 90 degree angle (the axial ones), with 2 at 120 degree angles. This shape makes an equilateral triangle with each side making a 120-degree angle. In an axial environment, there are 3 bonds/lp's at a 90 degree angle (the equatorial ones), with 1 at 180 degrees (the other axial one). The structure formed in the plane suggests that the molecular geometry of BF3 has the shape of trigonal planar (central atoms are surrounded by three-terminal atoms). In a basic trigonal bipyramidal structure, you've got 2 environments, an axial and an equatorial one: Therefore, to minimise the energy of the molecule, the lone pairs need putting in whichever position minimises their interactions. Therefore, molecule has a T-shape geometry.In a lone pair (lp), the average position of the electrons is closer to the central atom than the electrons involved in a covalent (in this case Cl-F) bond, so they have a slightly greater repulsion to other lone pairs (or bonds), than a bond does. Thus, F-Cl-F angle is a little less than 180°. Because repulsion involving lone pairs are stronger than bond pairs. The two lone pairs occupy equatorial position at an angle of 120° to each other, this gives the lowest energy arrangement of electron pairs in the molecule. 
These keep as far apart as possible, minimising repulsion between each of the negatively charged clouds by adopting a trigonal bipyramidal arrangement. The valence shell electron-pair repulsion theory (abbreviated VSEPR) is commonly used to predict molecular. Lewis structures are very useful in predicting the geometry of a molecule or ion. ClF3 Molecular Geometry ClF3 has a T-shaped molecular geometry and trigonal bipyramidal electron geometry. The valency of chlorine (Cl) is dependent on the other atoms to which it is bonded. As it is known that chlorine (Cl) can have more than one valency. Thus, ClF3 is T-shaped, having the axial FClF compressed from the angle of 180°, which is a formal sp3d hybridization. Three of these are bond pairs and two are lone pairs. Many of the physical and chemical properties of a molecule or ion are determined by its three-dimensional shape (or molecular geometry). ClF3 Polar or Nonpolar ClF3 is a polar molecule due to its asymmetrical structure and the presence of two lone pair electrons, which results in an unequal distribution of charge and so makes it polar. We can describe the molecular geometry in terms of atoms, but not electrons. The next highest repulsion is between one lone pair and a bond pair and the lowest is between two bond pairs.Īs applied to chlorine trifluoride, it results in a trigonal bipyramidal geometry for the shape-determining five electron pairs. 
As we know that the highest repulsion is between any two “lone electron pairs”, resulting in these moving apart as far as possible. Therefore, there are total 10 electrons or five electron pairs. Also, contribution of three fluorine atoms is 1 electron each. The standard application of VSEPR theory to Chlorine trifluoride is as follows: In chlorine trifluoride, central atom is chlorine and valence electrons on central atom is 7.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
